28th November 2025
Chemistry Society: Aspirin synthesis Winter Term 2025
During September and October, our Chemistry Society second-year students were learning about Carbonyl compounds in their lessons.
The synthesis was carried out over four weeks:
- Alkaline hydrolysis of the ester methyl-2-hydroxybenzoate. This made sodium-2-hydroxybenzoate, which, when acidified, produced the 2-hydroxybenzoic acid precursor molecule.
- Reacting 2-hydroxybenzoic acid with ethanoic anhydride to make the impure aspirin. Still some reactants/side products left, hence the need for further purification
- Purifying the aspirin by recrystallisation and testing its melting point to check purity.
- Titration of the aspirin by the back-titration method. In this final week, the first-year students joined us, and by then they had been trained to carry out an acid-base titration.
The process and chemistry are summarised in this video, which was made and provided for the students.

Above (L-R): A2 students Hannah, Amber, Lucy and Athena reflux their methyl-2-hydroxybenzoate using sodium hydroxide.

Above: a range of emotions can result from a 20 minute refluxing session.

Above: Hannah adds the ethanoic anhydride in the fume hood.

Above: testing the pH of the mixture after acidification.

Above: Zahra, Rachel and Insiyrah cool their mixture after recrystallising (below) to get the solid aspirin crystals to form.
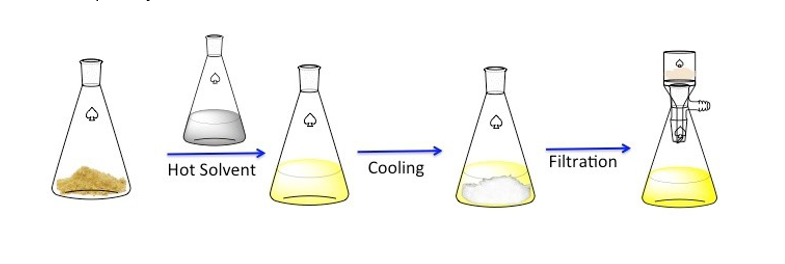

Above: Lucy and Athena test for the phenol functional group in 2-hydroxybenzoic acid (below) using iron(III) solution
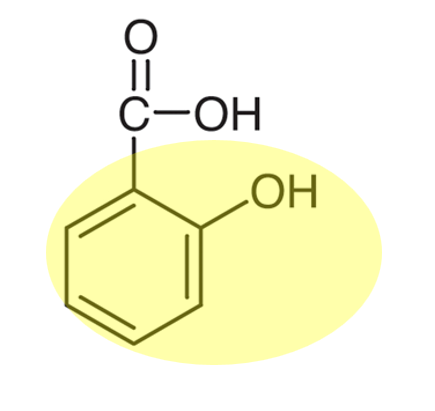

Above: The A-Team! A2 students Ava, Alex and Anna discuss whether their result proves there is still some 2-hydroxybenzoic acid left after testing for the phenol functional group.

Above: First year students Chris and Peter set up for the back titration

Above: First year students Lily and Grace prepare their aspirin sample for titrating.

Above: First year students Jeffrey and Oscar take their first measurement during the back-titration
