News
17th December 2019
17th December 2019
Extracting iron on a match head
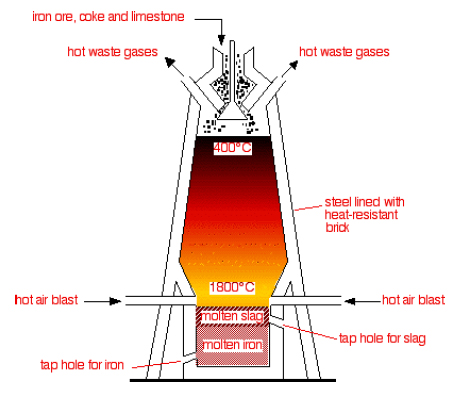
Angie, Jodie and Eleanor, all members of our Chemistry Society (ChemSoc), recently undertook a micro-scale reduction of iron (III) oxide, commonly known as haematite ore, or sometimes rust. By using a match as a source of carbon, they were able to replicate the inner workings of a blast furnace (below) using simple lab apparatus.
 |
 |
| Dampened match head is coated in iron (III) oxide | Match is ignited and carbon from the match is converted to carbon dioxide |
|
|
|
Click here for a pdf slideshow explaining the chemistry